Psychedelic drugs to treat mental health disorders has been around for decades, but in 2020 this area received the focus of investors with added relevance as the current pandemic develops into a global mental health crisis. However, there are social and legal barriers making the adoption of psychedelics a viable treatment option.
Subscribe to Dealroom’s Health newsletter, for weekly insights on healthtech, biotech and wellness:
Accelerating adoption
Psychedelics are a class of natural and synthetic compounds that includes psilocybin (the psychoactive ingredient in magic mushrooms), esketamine, MDMA, LSD, ibogaine and DMT. These are able to alter thoughts and sensory perceptions. Some psychedelics have been used by native communities for hundreds of years, and other psychedelics started being used in the middle 20th century by clinicians. In the late 90s, the DEA allowed researches from universities to study psychedelics in limited amounts (here more about its history).
According to Nature, in the last decade the number of clinical trials testing psychedelics to treat mental health conditions has increased from 3 in 2010 to 17 trials in 2020. The potential of these psychedelic drugs can treat depression, Post Traumatic Stress Disorder (PTSD), severe anxiety, substance abuse, anorexia nervosa and other disorders under close guidance of a psychiatrist or psychotherapist.
In May, it was published the results of the first Phase 3 clinical trial conducted with MDMA: “MDMA significantly attenuated PTSD symptomology”. While caution against possible long-term negative effects and abuse remain. There’s no research that has concluded that MDMA is addictive, but cases suggest that regular use of MDMA yields adaptations in the serotonin and dopamine pathways in the brain and central nervous system that may be related to hallucinogen use disorder.
Magic medicine
Since 2019, esketamine has been a hot area as Johnson & Johnson’s Spravato (branded name for esketamine) became the first FDA-approved psychedelic treatment for major depressive disorders with suicidal ideation or behaviour.
A study, published in April this year, evidences how psilocybin has rapid and sustained antidepressant effects in patients with major depressive disorders. In 2019, the FDA granted psilocybin “breakthrough” therapy designation. Psilocybin is not considered to be addictive, yet its chronic abuse and misuse can lead to substance use disorder.
From the possible adverse effects from the misuse of psychedelic drugs (such as euphoria episodes, dizziness, nausea, panic attacks and dissociation from reality), their illegality still makes them dangerous to be regulated. Nevertheless, its use is promising for the treatment of some of the most difficult psychiatric conditions to be treated, such as PTSD. For this, regulators are establishing a strict set of protocols for psychedelic drugs to avoid misuse. For instance, patients being treated with psilocybin can only consume psychedelic drugs at a licensed space with a certified professional present.
Funding trends
Global VC funding in psychedelic drugs has gone from €3 million in 2016 to €399 million 2021YTD, a 130x increase. The largest rounds, so far, has been for the German ATAI Life Sciences ($157 million Series D and $125M Series C), followed by COMPASS Pathways $80M Series B.
We have identified and analysed 30 Biotech startups developing psychedelics drugs.
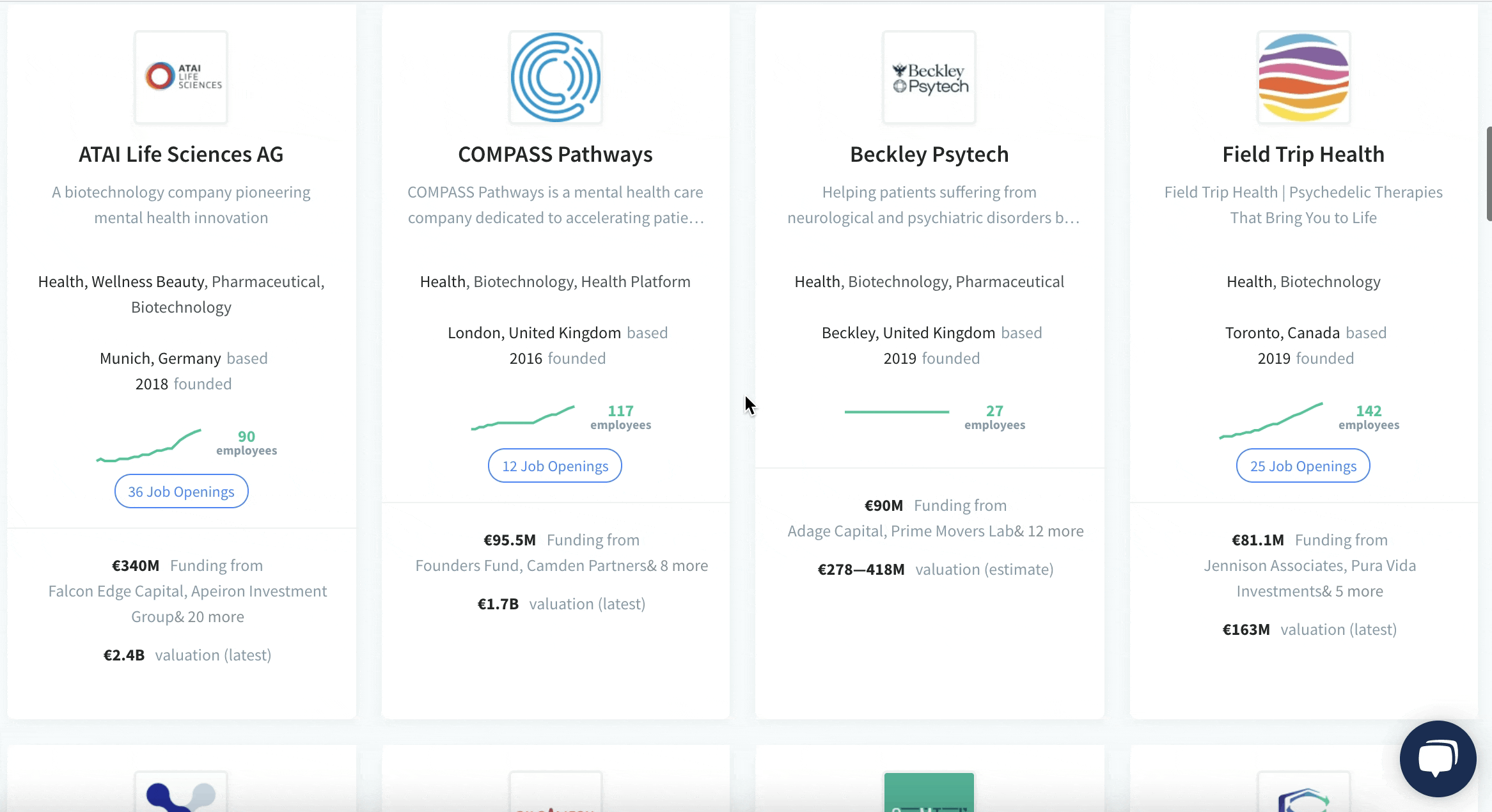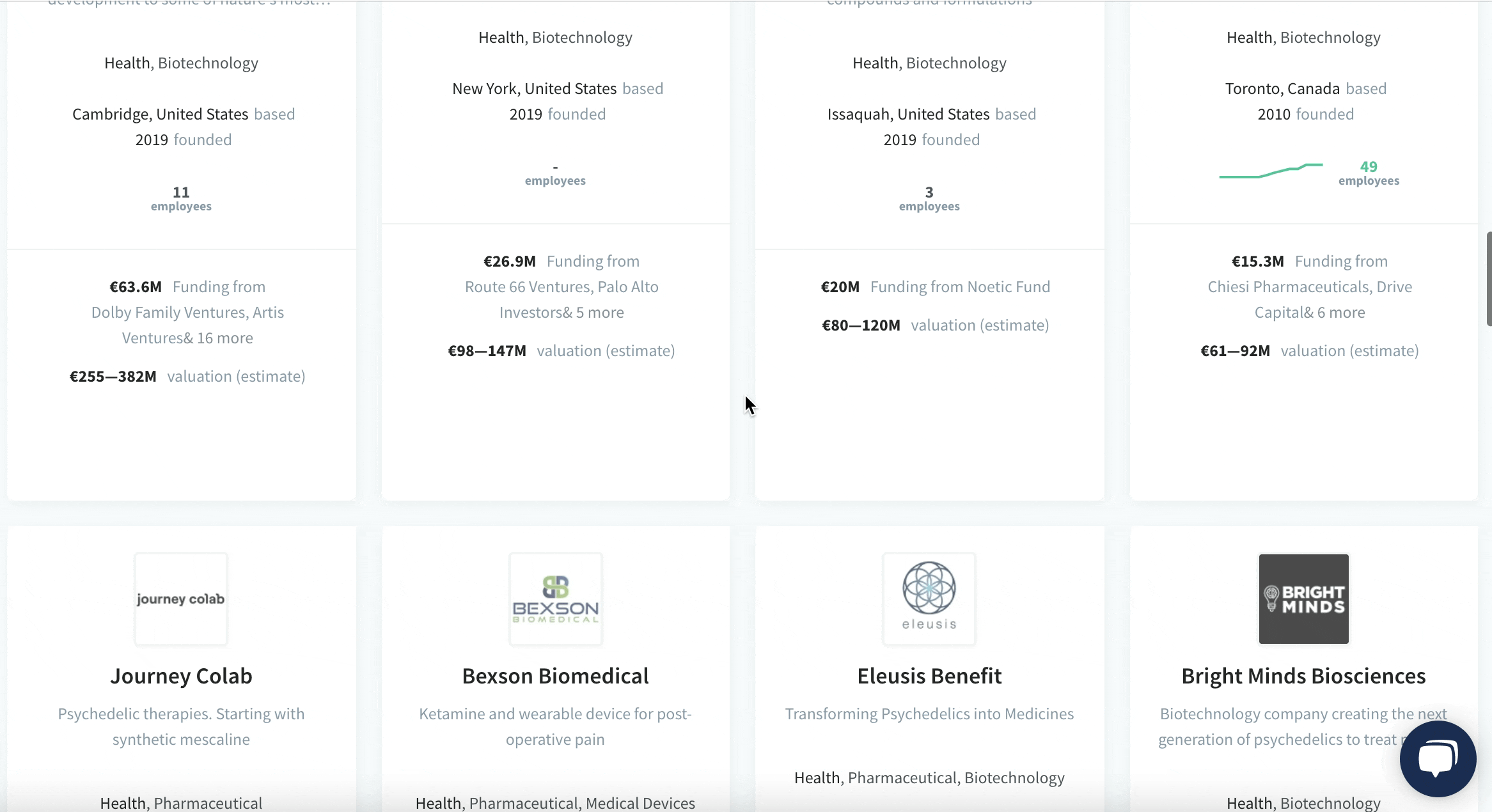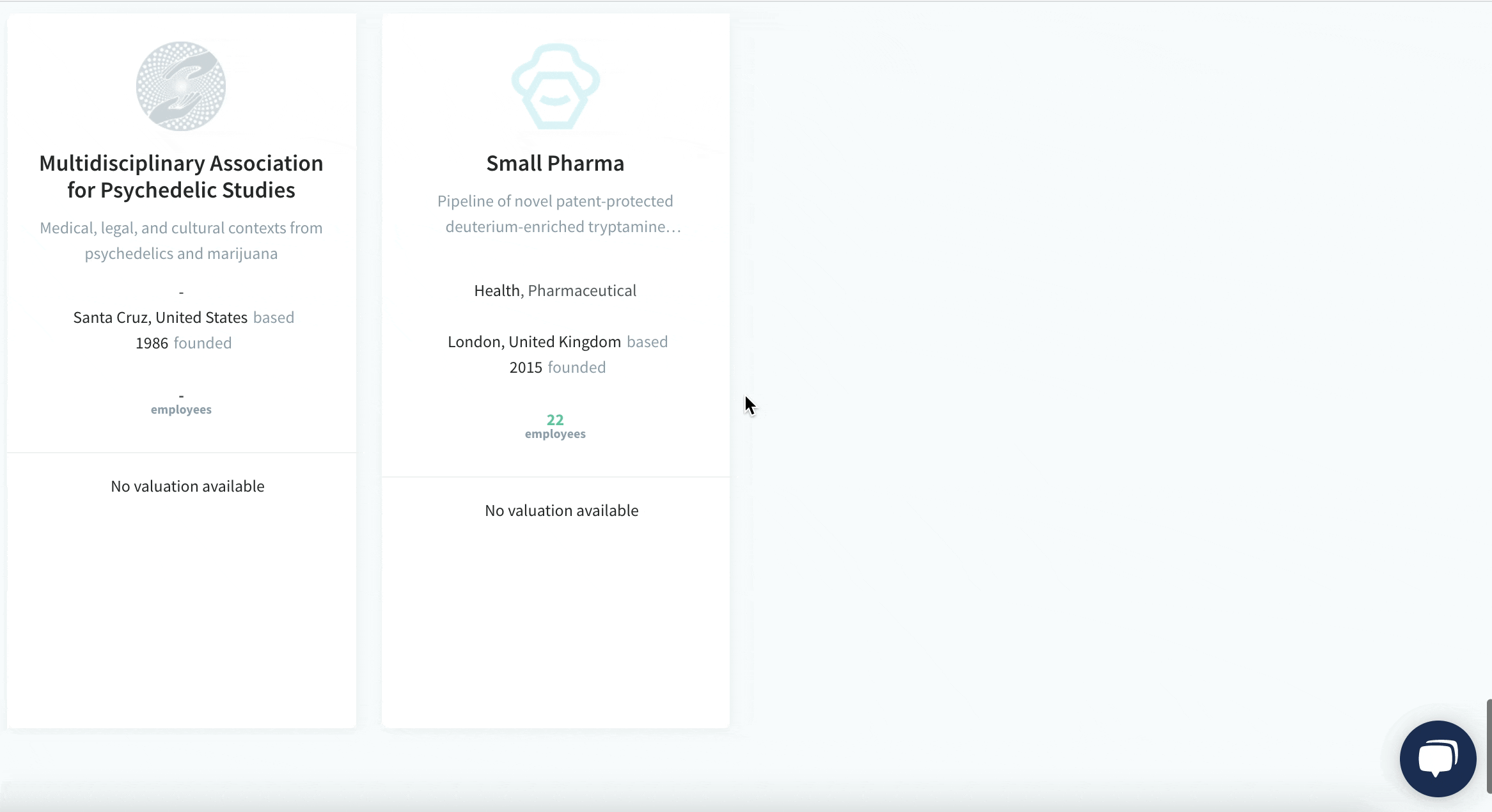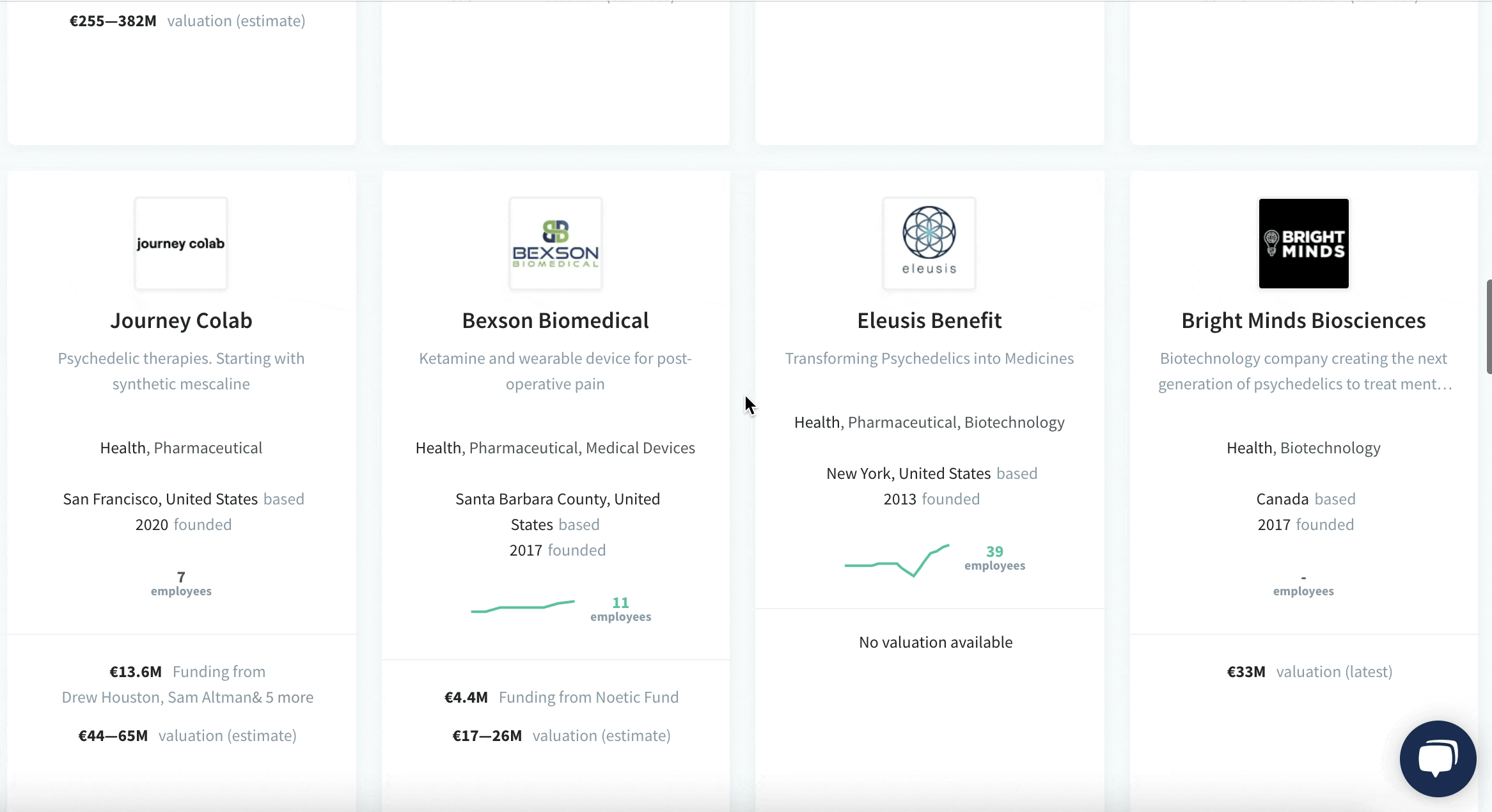
The pioneering European Biotech startups developing medicines from psychedelics and its milestones:
🇩🇪 ATAI Life Sciences, clinical-stage biopharmaceutical company focused on the treatment of mental health disorders using psychedelic drugs (psilocybin, ketamine, ibogaine, MDMA). ATAI invested in COMPASS Pathways and has also created joint ventures (programs) with others in the space such as Perception Neuroscience, Recognify Life Sciences, DemeRX, and more.
ATAI went public in June 2021, and now it’s valued at $2.3 billion. Its stock was up more than 20% last week, outperforming Tesla, Nvidia and GameStop. So far, it’s the most valuable Biotech startup developing psychedelics drugs.
🇬🇧 COMPASS Pathways, is currently working on the world’s largest trial, Phase IIb trial of COMP360, its proprietary formulation of psilocybin being tested to treat depression across Europe and North America. The company went public in Sep 2020, and now it’s valued at $1.9 billion. Its stock has fallen 27% from its December record high.
🇬🇧 Beckley Psytech, clinical stage company dedicated to treat neurological and psychiatric disorders by developing a broad pipeline of psychedelic compounds in rare and common diseases.
Beckley Psytech announced last week first cohort dosed in Phase I clinical trial assessing safety and tolerability of intranasal 5-MeO-DMT (psychedelic found in a wide variety of plant species, and also is secreted by the glands of the Sonoran Desert toad) in psychedelic-naïve healthy subjects. This is the first clinical study to measure the pharmacokinetics and metabolism of 5-MeO-DMT delivered intranasally.
See the full list of 30 Biotech startups developing psychedelics drugs.
Subscribe to Dealroom’s Health newsletter, for weekly insights on healthtech, biotech and wellness:

