Digital therapeutics: medical intervention “beyond the pill”?
Digital therapeutics (DTx) are playing an increasingly central role in the future of healthcare, by delivering an evidence-based intervention via software or medical device to replace or complement disease treatment. DTx influences patients’ behaviour without using a pill (medication-free treatment).
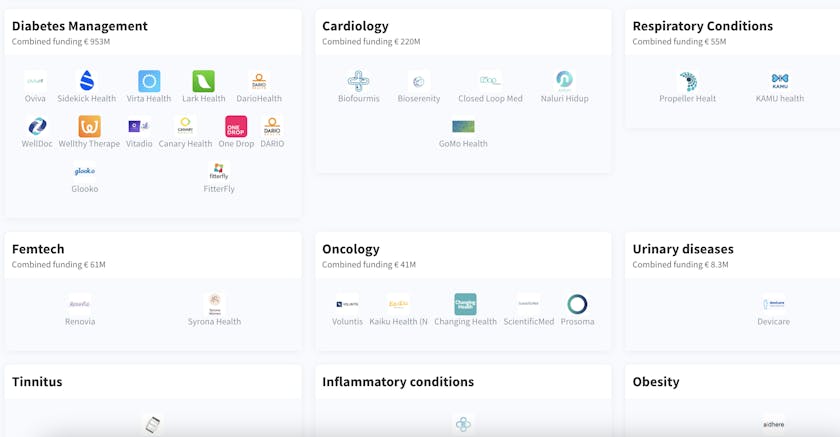
DTx focuses on a range of medical conditions and supports various stages of the traditional healthcare process. These products go from tracking and managing symptoms, to delivering cognitive behavioural therapy and treatments for musculoskeletal and gastrointestinal conditions. We have identified and analysed 100+ DTx startups.

Digital therapeutics regulations
DTx differentiate from digital health products, as DTx must be approved by regulators. They must follow the standards of any other medical product and treatment. These need to go through clinical trials to demonstrate safety, privacy, quality and efficacy. The necessity for clinical evidence helps to demonstrate the therapeutic efficacy done by the software and/or hardware, but also the possible reimbursement by insurers and prescription by professionals.
DTx is still a young health sector, facing regulatory and payer-side challenges. About 35+ DTx have been approved by the FDA since 2017, but so far, the FDA doesn’t have a specific definition for DTx. However, during the pandemic, the FDA has increased flexibility so digital health tools and DTx can become more accessible.
Digital therapeutics market
As DTx are evidence-based and regulated, compared to traditional experimental drugs, DTx cost significantly less in research and clinical trials (McKinsey), making them more attractive and easier to implement at a lower risk.
The utility of digital therapeutics is being at the attention of all stakeholders in the healthcare system. Patients want convenient and informative digital products, but making it accessible depends also on socioeconomic factors. Pharmaceuticals are seeking to develop more digital solutions and are getting involved in DTx platforms, not to replace drugs, but to improve outcomes with both (Bozidar Jovicevic, Sanofi). Providers and physicians need well integrated digital solutions that can help them to provide to more patients a more effective service at a lower cost. The regulatory bodies are already working towards digital innovation in healthcare.
DTx are products that are reimbursed as payers value a therapy based on its evidence to reduce costs and complications. At a national level, Germany was the first European country to reimburse prescription digital therapeutics: e.g. Kalmeda (tinnitus), Velibra (anxiety) and recently, HelloBetter (mental health) can now be prescribed to around 50 million adults in Germany. In the UK, reimbursement is in a regional level. And in France, Emmanuel Macron announced plans to replicate Germany’s DiGA Fast Track process. In the US, DTx receive third-party payment through employers, Pharmacy Benefit Managers (PBM), insurers, or other third-party payers.
Digital therapeutics funding trends
VC funding in DTx has gone from €202 million in 2016 to €1.6 billion in 2021YTD, a 8x increase. The increase has been led by megarounds (such as those for Hinge Health, Virta Health, Akili, Vida Health and Lark Health). But the early-stage scene with rounds below €100 million is also booming with funding from SWORD Health, Kaia Health, Happify Health and DarioHealth.
The vast majority of the investment is directed towards North American firms, with European startups gaining a bit more traction only in the past two years.
The pioneering European startups tackling DTx:
🇩🇪 Kaia Health, AI-assisted digital therapies via a mobile app for chronic pain related to musculoskeletal (MSK) disorders and for Chronic Obstructive Pulmonary Disease (COPD).
🇬🇧 Ieso Digital Health, AI-based company offering cognitive behavioural therapy via text-based messaging that takes place in a secure virtual room.
🇸🇪 Joint Academy delivers chronic joint pain treatment online. It consists of daily exercises via videos, tools to track progress, and patient education depending on their condition.
🇫🇷 Voluntis, provides real-time, personalized recommendations to patients, so they know what to do and how to manage their symptoms remotely. DTx in oncology, diabetes and coagulation.
🇩🇪 ATAI Life Sciences launched in 2020 IntroSpect Digital Therapeutics, a DTx program to test the digital approach in patients receiving ketamine for treatment resistant depression.
Interested?